The article covers the types of fossil fuels (coal, oil, and natural gas), their formation, uses, and the environmental impact of their consumption. It also briefly discusses renewable energy sources, the inefficiencies in energy generation and distribution, and the role of greenhouse gases in global warming.
Fossil Fuels
Fossil fuels are fuels formed from decaying plant and animal matter that was primarily formed over millions of years. Fossil fuels include coal, oil (petroleum), and natural gas. These fuels have been the primary energy source for over 200 years, but they will eventually have to be replaced by other sources because the world supply of fossil fuels is finite. The amount of each type of fossil fuel that is left is debated, but it is clear that the continued rate of use for any fossil fuel cannot be sustained. Today, fossil fuels supply approximately 80% of the energy consumed in the United States and about 87% worldwide according to the BP Statistical Review of World Energy.
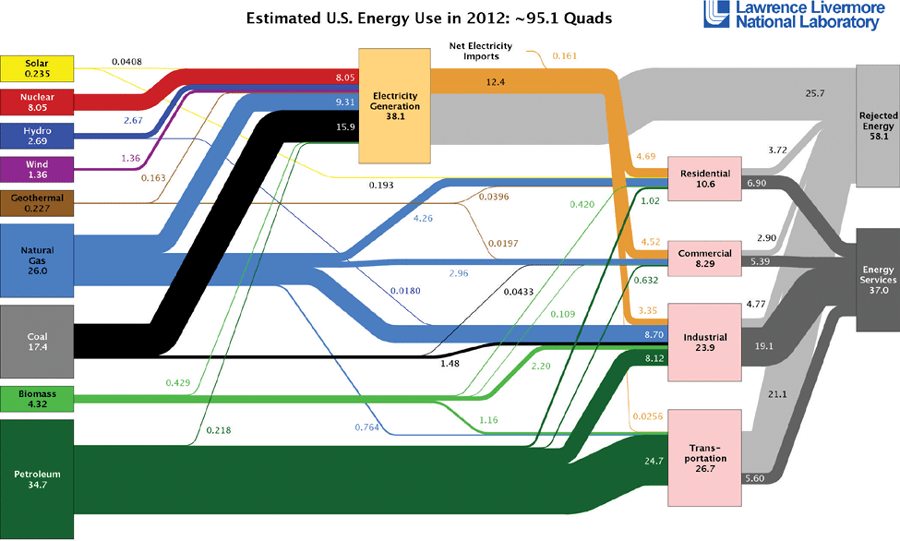
An interesting tool for visualizing the sources of energy and ultimate outcome is the so-called spaghetti diagram, which was developed and used by Lawrence Livermore National Laboratory. Figure 1shows the diagram with line width proportional to the percentage of each source; this one is for a recent year in the United States. It clearly shows the dependency on fossil fuels. The left side shows the various sources and the right side shows the demand sectors. Electricity is an intermediate step and not a demand in itself. Notice that the largest source of energy comes from petroleum. Approximately 72% of the petroleum is used in the transportation sector. In the case of electricity, 48% of electrical energy comes from coal-fired plants and, as you can see, the process creates a lot of rejected heat. Rejected heat is due to the inefficiencies in generating and distributing electrical power. Notice that electricity production is the primary user of coal. The best electrical generation plants are less than 50% efficient, and 5 to 10% of the energy is lost in transmission lines. It is important to be aware of the inefficiencies in the generating and distribution process because it affects other demand areas.
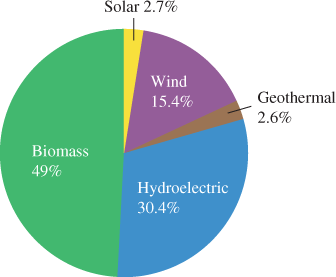
Renewable energy sources—biomass, hydroelectric, geothermal, wind, and solar— constitute only a small percentage of the overall mix at this time. All are covered in more detail throughout this text, but a quick overview of the current mix of renewable sources in the United States is shown in Figure 2. The mix has been changing rapidly in recent years, with significantly more wind energy. Biomass, the largest source, includes a variety of sources, including wood, waste, garbage, and even plants that are grown for fuel. Hydroelectric is the second largest source today because of the huge infrastructure of dams and power plants. Other sources (wind, geothermal, and solar) account for 14% of renewable use in the United States.
Worldwide consumption is continuing to rise for all fossil fuels. Fossil fuels include coal, petroleum and natural gas.
Figure 1:Energy Flow for the United States
Coal
Coal has been used as a fuel for more than 2,000 years, and historical records document its usefulness to the early Greeks, Romans, and Chinese as well as other cultures. Large-scale mining of coal was brought on by the Industrial Revolution with the development of the steam engine and improvements in steel making. Coal is a combustible rock that is composed mostly of carbon and hydrocarbons. A hydrocarbon is a molecule containing only hydrogen and carbon (but not all hydrocarbons are fuels). Coal is derived from ancient plant life, mainly trees. It is believed that these ancient terrestrial forests were flooded rapidly and eventually sank, where layer upon layer of dying plants was covered by sediments. Mild heat and pressure condensed the organic material into peat in a process called digenesis. If enough heat and pressure is supplied, the organic material will undergo physical and chemical change to form coal. This process takes several million years and turns the peat gradually into coal, which is found in sedimentary layers. Figure 3 shows a coal seam in a sedimentary rock formation.
Figure 2:Renewable Energy Sources in the United States
Figure 3: A Coal Seam in Sedimentary Rock
Depending on the conditions and the amount of carbon in the original materials, different types of coal formed. Coal is classified into four main categories based on energy content: anthracite, bituminous, sub-bituminous, and lignite (anthracite has the highest energy, but is not as common as the other types.). Most coal today is used for generating electricity, but a smaller percentage is used by industry for making steel and other products.
Petroleum (Oil)
Petroleum is also a nonrenewable fossil fuel that formed in the distant past in a two-step process. The process starts when aquatic organic sediments are compacted and when heat and temperature break it down with the aid of microbes into a waxy material known as kerogen and a black tarlike hydrocarbon called bitumen. Bitumen can occur naturally or as a product of refining petroleum. Kerogen can undergo further chemical and physical change in a process called catagenesis if it is compacted and buried deeper underground where temperatures and pressures are higher. In this case, water is squeezed out and the kerogen breaks down into hydrocarbon chains by a process that is aided by the presence of certain minerals in marine deposits. This is equivalent to cracking, a term used by refineries when crude oil is converted to gasoline and other products. At the highest temperatures, natural gas forms. If the temperature is lower, oil forms. If the temperature is lower still, the kerogen remains unaltered. Carbon, with four electrons in its outer shell, has the ability to bond to other carbon atoms and form long chains and complex atomic arrangements with hydrogen and is the fundamental chemical structure in both petroleum and natural gas.
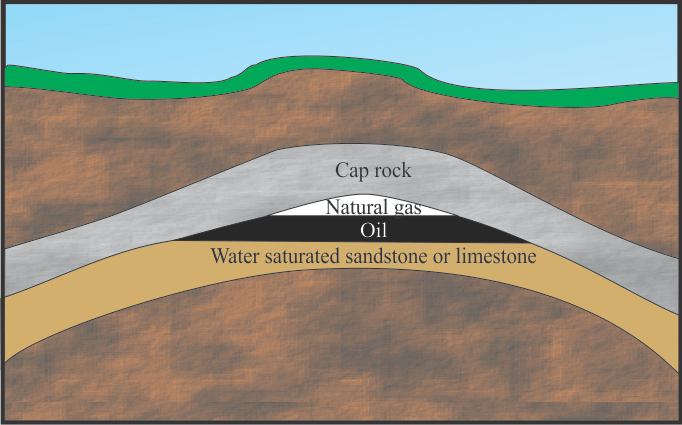
The density of the oil and natural gas is lower than the rock layers in which it is buried, so these substances would normally migrate to the surface. Instead, they are trapped by a layer of impervious rock called cap rock, which is typically shale. The cap rock traps the gas and oil in porous sedimentary rock formations. Large volumes of natural gas and viscous liquid oil are trapped in these underground regions called reservoirs. The natural gas is under pressure and escapes when the formation is drilled. Figure 4 illustrates how oil and gas and sometimes water are trapped underground by the cap rock. There are various types of oil and natural gas traps, but the common feature is that oil moves through the porous rock layer and is trapped by an impervious layer in the underground reservoir. Reservoirs that contain very hot water under pressure are useful as a geothermal heat source.
Figure 4: Underground Reservoir. The particular reservoir shown is an anticline. Other types have been identified by geologists.
The principal use for petroleum is in the transportation sector. In the United States, approximately 72% of all petroleum is used to make gasoline, diesel, and other products for vehicles. One important use for petroleum that is often overlooked is in chemical feed stock. It is also used in the manufacture of many products including lubricants, waxes, solvents, asphalt, hydraulic fluid, and vinyl, to name a few.
Carbon Dioxide and Methane
Carbon dioxide emissions result from burning of fossil fuels and other fuels, as well as certain other chemical reactions such as those during production of cement. An example of a naturally occurring emission is volcanic eruption.
Methane enters the atmosphere primarily from the production of fossil fuels (coal, oil, and natural gas), livestock, and the decay of organic waste. The Environmental Protection Agency (EPA) estimates that one-half of all methane enters the atmosphere from human activities.
Natural Gas
The primary constituent of natural gas is methane, the simplest hydrocarbon. Although natural gas is nonrenewable, methane, a principal component of natural gas, can be produced by various processes such as the decomposition of waste in landfills and by anaerobic (without oxygen) decay of organic matter such as manure or biomass. The chemical formula for methane is CH4. This formula indicates a single carbon atom is bound to four hydrogen atoms. When methane burns, it reacts with oxygen in the air to release energy and form carbon diode and water as products. This basic chemical reaction for burning methane is:
CH4+2O2→CO2+2H2O+energy
The reactants (methane and oxygen) are written on the left side and the products of the reaction (carbon dioxide and water) are written on the right side. This equation not only shows the reactants and products, it also shows the ratios of molecules involved. For each molecule of methane, two molecules of oxygen combine to release one molecule of carbon dioxide and two molecules of water plus energy. As shown by the chemical equation, a by-product of this reaction is carbon dioxide, which typically escapes into the atmosphere as a by-product of combustion. In addition to methane, natural gas also contains other, more complex hydrocarbons as well as some other undesirable materials, including sulfur. Carbon dioxide and methane are each considered to be a greenhouse gas. Greenhouse gases contribute to the greenhouse effect by absorbing short wavelength infrared energy and reradiating it at longer wavelengths. Other greenhouse gases include nitrous oxide (NO2), fluorinated gases (CFC, etc.), and water vapor (H2O).
From the chemical formula for the reaction of burning methane, the ratio of masses of the reactants and products can easily be determined by determining their molecular weights. The atomic weights are given on the periodic table of the elements and other sources. In a chemical reaction, mass is always conserved; that is, the mass of reactants is equal to the mass of the products. To determine the relative weight of the reactants and products, look up the atomic weights of each atom, then determine molecular weights by multiplying the number of atoms of each type by its atomic weight. The following example illustrates the idea.
Example
What is the mass of carbon dioxide (CO2) that is released to the atmosphere if 1,000 kg of methane (CH4) is burned? (One thousand kg is approximately 2,200 pounds, or 1.1 tons.)
Solution
CH4+2O2→CO2+2H2O+energy
The equation shows that each molecule of CH4 reacts with 2 molecules of O2 to produce 1 molecule of CO2 and 2 molecules of H2O. Start by determining the molecular weight of each reactant and product. By expressing the molecular weight in grams, you will obtain the relative masses of the reactants and products. The atomic weight of carbon is 12.0 g, hydrogen is 1.0 g, and oxygen is 16.0 g.
CH4 = 1 carbon and 4 hydrogen = 1(12.0 g) = 16.0 g
2O2 = 2 molecules of oxygen, each with 2 atoms = 4(16.0 g) = 64 g
CO2 = 1 carbon and 2 oxygen = 1(12.0 g) + 2(16.0 g) = 44.0 g
2H2O = 2 water molecules each with 2 hydrogen and 1 oxygen = 4(1.0 g) + 2(16.0 g) = 36 g
Thus, 16 g of CH4 reacts with 64.0 g of O2 to produce 44 g of CO2 and 36 g of H2O You can check that the mass on each side is the same, as it must always be in chemical reactions. From this you can set up a proportion:
(16 g CH4) / (1, 000 kg CH4) = (44 g CO2)/ X kg CO2
Solving for X (the unknown quantity of CO2), gives X = 2,750 kg CO2 which is approximately 3 tons. This illustrates that burning methane creates a weight of carbon dioxide that is 2.75 times the weight of the original methane gas
Applications for natural gas include heating for homes and businesses. Most of the heat from the reaction is available for heating; a small fraction escapes with flue gases. Natural gas is also used as an alternative to gasoline for automobiles and trucks and is widely used in electrical power generation. As an electrical power energy source, it emits the lowest amount of carbon dioxide of any fossil fuel per unit of energy produced. Natural gas turbines are used to supplement renewable sources when they are not available because natural gas turbines have quick start-up times and thus can be brought on line rapidly. Another natural gas application is as a fuel source for some fuel cells; it may find more widespread use when fuel cell vehicles become more common in the future.
Types of Fossil Fuels Key Takeaways
The understanding of fossil fuels and their applications is crucial in addressing energy needs and the environmental challenges that come with their consumption. Fossil fuels, such as coal, oil, and natural gas, have been the dominant sources of energy for centuries, powering industries, transportation, and electricity generation. However, their finite nature and harmful environmental impact, including greenhouse gas emissions and global warming, underscore the need for sustainable alternatives. While renewable energy sources like wind, solar, and geothermal show promise in reducing reliance on fossil fuels, inefficiencies in energy generation and distribution still present challenges. It is important to continue advancing renewable energy technologies and optimizing the use of fossil fuels in ways that mitigate environmental damage. Recognizing these energy sources’ role in applications like electricity generation, heating, and transportation helps inform decisions that balance energy demand with long-term sustainability goals.