These three basic electrical quantities—energy, charge, and voltage—are closely related. It is difficult to visualize or measure energy directly because it is an abstract quantity and represents the ability to do work. The electrical charge can be positive or negative, and it can do work when it moves from a point of higher potential to one of lower potential. Voltage is a measure of the energy per unit of charge and can be measured easily with common instruments. Voltage is one of the electrical quantities that you will work within most renewable energy systems.
Energy
Work is done whenever an object is moved by applying a force over some distance. To do work, you must supply energy.
Energy is the ability or capacity for doing work; it comes in three major forms; potential, kinetic, and rest.
Stored energy is called potential energy and is the result of work having been done to put the object in that position or in a configuration such as a compressed gas. For example, the water stored behind a dam has stored (potential) energy because of its position in a gravitational field.
Kinetic energy is the ability to do work because of motion. The moving matter can be a gas, a liquid, or a solid. For example, the wind is gas in motion. Falling water is a liquid in motion; a moving turbine is solid in motion. Each of these processes is a form of kinetic energy because of the motion.
Rest energy is the equivalent energy of matter because it has mass. Einstein, in his famous equation E = mc2, showed that mass and energy are equivalent.
Unit of Energy
Because energy is the ability to do work, energy and work are measured in the same units. In all scientific work, the International System of Units (SI) is used. SI stands for Système International, from French. These units are the basic units of the metric system.
Energy, force, and many other units are derived units in the SI standard, which means they can be expressed as a combination of the seven fundamental units. The most common derived units are those using three fundamental units, which are the meter, kilogram, and second (mks). This forms the basis of the mks system of units, which are the most common derived units in the SI system. Another derived set of units is based on the centimeter, gram, and second (cgs). These smaller units are referred to as the cgs system.
The SI unit for energy is the joule (J), which is defined to be the work done when 1 newton of mechanical force is applied over a distance of 1 meter. A Newton is a small unit of force, equivalent to only 0.225 pounds. The symbol W is used for energy, and we will use WPE or WKE to specify potential energy and kinetic energy, respectively, to be consistent with W. (You may see E for energy in some cases, such as Einstein’s E = mc2 or PE and KE for potential energy and kinetic energy, respectively). The equation for gravitational potential energy is
${{W}_{PE}}=mgh$
Where
WPE = potential energy in J
m = mass in kg
h = height in m
The equation for kinetic energy is
${{W}_{KE}}=\frac{1}{2}m{{v}^{2}}$
Where
WKE = kinetic energy in J
m = mass in kg
v = velocity in m/s
Electrical Charge
Charles Augustus Coulomb (1736–1806) was the first to measure the electrical forces of attraction and repulsion of static electricity. Coulomb formulated the basic law that bears his name and states that the force between two point charges is proportional to the product of the charges and inversely proportional to the square of the distance between them. His name was also given to the unit of charge, the coulomb (C).
Coulomb’s law works for like charges or unlike charges. If the signs (+ or −) of both charges are the same, the force is repulsive; if the signs are different, the force is attractive. Long after Coulomb’s work with static electricity, J. J. Thomson, an English physicist, discovered the electron and found that it carried a negative charge.
The electron is the basic atomic particle that accounts for the flow of charge in solid conductors. The charge on the electron is very, very tiny, so literally many trillions of electrons are involved in practical electrical circuits. The charge on an electron was first measured by Robert Millikan, an American physicist, and found to be only 1.60 × 10−19 C. The power of ten, 10−19 means that the decimal point is moved back 19 decimal places.
Voltage
Voltage (V) is defined as energy (W) per unit charge (Q). The volt is the unit of voltage symbolized by V. For example, a battery may produce twelve volts, expressed as 12 V. The basic formula for voltage is
$V={}^{W}/{}_{Q}$
One volt is the potential difference between two points when one joule of energy is required to move one coulomb of charge from one point to another.
Sources of Voltage
Various sources supply voltage, such as a photovoltaic (solar) cell, a battery, a generator, and a fuel cell, as shown in Figure 1. Huge arrays of solar modules can provide significant power for supplying electricity to the grid.
Figure 1 Sources of Voltage
Figure 2 DC Voltage Source
DC Voltage
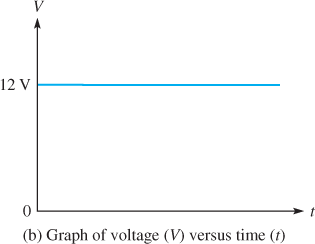
Voltage is always measured between two points in an electrical circuit. Many types of voltage sources produce a steady voltage, called dc or direct current voltage, which has a fixed polarity and a constant value. One point always has positive polarity and the other always has negative polarity. For example, a battery produces a dc voltage between two terminals, with one terminal positive and the other negative, as shown in Figure 2(a). Figure 2(b) shows a graph of the ideal voltage over time. Figure 2(c) shows a battery symbol. In practice, the battery voltage decreases some over time. Solar cells and fuel cells also produce the DC voltage.
AC Voltage
Electric utility companies provide a voltage that changes direction or alternates back and forth between positive and negative polarities with a certain pattern. AC generators produce the alternating voltage or alternating current (ac) voltage. In one cycle of the voltage pattern, the voltage goes from zero to a positive peak, back to zero, to a negative peak, and back to zero. One cycle consists of a positive and a negative alternation (half-cycle). The cyclic pattern of ac voltage is called a sinusoidal wave (or sine wave) because it has the same shape as the trigonometric sine function in mathematics.
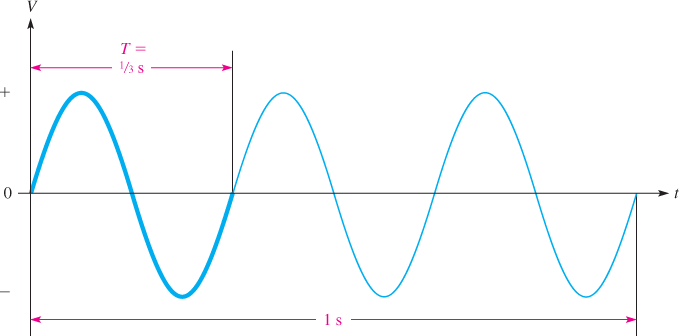
In North America, ac voltage alternates one complete cycle 60 times per second; in most other parts of the world, it is 50 times per second. The number of complete cycles that occur in one second is known as the frequency (f).Frequency is measured in units of hertz (Hz), named for Heinrich Hertz, a German physicist. Figure 3 illustrates the definition of frequency for the case of three cycles in one second, or 3 Hz.
Figure 3 Example of an AC Sinusoidal Voltage. The frequency is 3 Hz, and the period (T) is ⅓ s.
The period ( T ) of a sine wave is the time required for 1 cycle. For example, if there are 3 cycles in one second, each cycle takes one-third second. This is illustrated in Figure 3, where one cycle is shown with a heavier curve. From this definition, you can see that there is a simple relationship between frequency and period, which is expressed by the following formulas:
$f=\frac{1}{T}$
$T=\frac{1}{f}$
EXAMPLE 1
- What is the voltage if the energy available for each coulomb of charge is 100 J and the total charge is 5 C?
Solution
\[V=W/Q=100J/5C=20\,V\]
- If the period of an ac voltage 0.01 s, determine the frequency.
Solution
\[f=1/T=1/0.01s=100Hz\]
- If the frequency of an ac voltage is 60 Hz, determine the period.
Solution
\[T=1/f=1/60Hz=0.0167s=16.7ms\]
Review Questions
- What is energy, and what is its unit?
- What is the smallest particle of negative electrical charge?
- What is the unit of electrical charge?
- What is voltage, and what is its unit?
- Name two types of voltage.
- Define frequency and period.
Answers
- Energy is the ability or capacity for doing work; it is measured in joules in the SI system.
- The electron
- The coulomb
- Energy per charge; the unit is the volt, symbolized by V.
- DC voltage and AC voltage
- Frequency is the number of cycles per second measured in Hertz. The period is the time for one cycle, measured in seconds.
Energy Charge and Voltage Key Takeaways
Understanding the fundamental electrical quantities—energy, charge, and voltage—is essential for grasping the principles that power nearly every modern electrical and renewable energy system. These concepts are deeply interconnected: energy represents the capacity to do work, charge is the carrier of energy in electric circuits, and voltage is the measure that tells us how much energy is available per unit of charge. Their applications span a wide range—from the operation of simple batteries and household electronics to complex systems like solar panels, electric vehicles, and grid-level energy infrastructure.